
Search your desired product with Calgan
Latest Collection
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
- Buy via WhatsApp
Featured Collection
Calif D Tablets
FERONA LIQUID
FECTO LIQUID
PRIMAL-DK DROPS
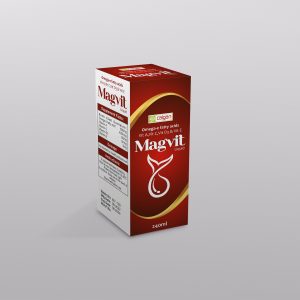
MAGVIT IQUID
APIFOL TABLET
ADIPEX TABLET
XEMCO 10 TABLET
GLYCIFOL TABLET
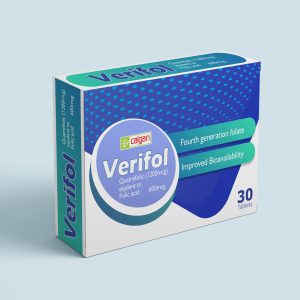
VERIFOL TABLET
ARQ-E-SHIRIN
CALPAN –D TABLET
SA TONIC
TREBLOGIN TABLET
COCALC TABLET
L-GEN PLUS SACHET
XANO PLUS TABLETS
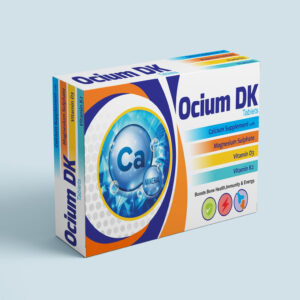
OCIUM DK
PEPRO COLIC LIQUID
DEMORE ORAL DROPS
QULET TABLETS
XINBAL TABLET
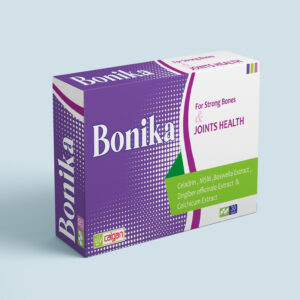
BONIKA TABLET
RENUMENT TABLET
EVOTOL TABLET
GAMMET-S TABLET
GYNOFOL TABLET
CALMIN-D TABLET
CYNORIVE TABLET
ABORAX TABLET
We have a state-of-the-art cGMP compliant manufacturing facilities with large capacities for manufacturing tablets, capsules, syrups, suspensions, ointments, creams,gells,Food supplements , Sachet and Phyto Chemicals
With over two decades of serving your healthcare needs , a vision to enhance the quality of life, Calgan Phyto Pharma is one of the leading Pharmaceutical companies, committed to providing you with best healthcare solutions. We develop and manufacture pharmaceutical formulations through our dedicated R&D team and state-of-the-art manufacturing facilities to ensure not only a healthier today but also, a healthier tomorrow!
Calgan aims at providing high-class medicines with greater access for the benefit of everyone. The company dedicatedly works towards improving the quality of human life through continuous innovation and hard-work to bring forth a stream of medicinal products that contribute to the betterment of the society.Calgan offers a broad portfolio of medicines seeking to give maximum benefit to patients in terms of Quality, Efficacious and economical medicine products. Among the medical professionals and other stakeholders, Calgan is best known for its dedicated people, passion for innovation through research and development and quality healthcare solutions. Determined to always go beyond its reputation , the recent development of its new GMP state-of-the-art manufacturing facility is a testimony of our continuous efforts to bring Calgan to the global fore.
With a diligent team and a cGMP compliant, manufacturing facility equipped with latest technology, Calgan is devoted on ensuring that it delivers and fulfils its commitments to the fullest. Strictly adhering to international maintenance and monitoring standards and stringent quality control checks, the company has established an edge over its competitors enabling it to become the industry benchmark.






Customers reviews